The global iron supplement market is undergoing a seismic shift. For decades, Ferrous Sulfate has been the industry standard due to its low cost. However, for R&D professionals and procurement managers, the hidden costs of traditional iron—low patient compliance, gastrointestinal side effects, and poor bioavailability—are becoming impossible to ignore.
As consumer demand moves toward "high-efficacy, low-irritation" products, Liposomal Iron has emerged as a superior alternative to traditional ferrous sulfate, especially with the growing demand for high-performance liposomal iron solutions. At LiposoMore, we are at the forefront of this transition, providing a delivery system that redefines how iron is absorbed and tolerated, while delivering proven liposomal iron benefits.
The Bioavailability Challenge: Why Ferrous Sulfate Falls Short
Traditional iron salts like Ferrous Sulfate rely on a precarious absorption pathway in the duodenum. Once ingested, they often lead to:
Oxidative Stress: Unabsorbed iron in the gut promotes the formation of free radicals.
Gastrointestinal (GI) Distress: Up to 40% of patients report nausea, constipation, or metallic aftertaste, leading to high dropout rates.
Low Net Absorption: Due to dietary inhibitors (like phytates or tannins), the actual amount of iron that reaches the bloodstream is often negligible, highlighting the limitations compared to modern liposomal iron supplement solutions.
The LiposoMore Advantage: A Superior Delivery Mechanism
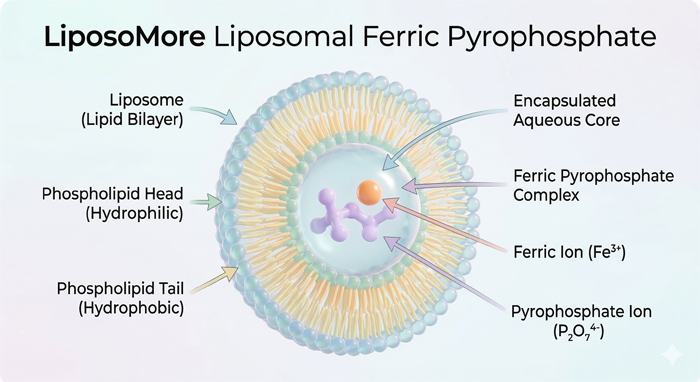
Liposomal Iron utilizes a sophisticated delivery system where the liposomal iron pyrophosphate is encapsulated within a phospholipid bilayer. This spherical vesicle mimics human cell membranes, offering several technical advantages for formulators:
1. Enhanced Absorption via Alternative Pathways
Unlike Ferrous Sulfate, which requires specific transporters, liposomal iron can be absorbed via endocytosis and the lymphatic system. This bypasses the typical "gatekeepers" of iron absorption, significantly increasing serum ferritin levels.
2. Gastric Protection and Reduced Side Effects
Because the iron is "shielded" inside the liposome, it does not come into direct contact with the gastric mucosa. This virtually eliminates the stomach irritation and metallic taste associated with traditional salts.
3. Protection from Dietary Inhibitors
The phospholipid shield protects the iron molecules from binding with dietary components like calcium or caffeine, ensuring consistent dosage regardless of the consumer's diet.
Technical Comparison: Liposomal Iron vs. Ferrous Sulfate
Feature | Ferrous Sulfate | Liposomal Iron (LiposoMore) |
Bioavailability | Low (approx. 10-15% ) | High (up to 3.5-5.0 times higher) |
GI Tolerance | Poor (Nausea, Constipation) | Excellent (Non-irritating) |
Organoleptic Profile | Strong Metallic Taste | Neutral / Clean |
Formulation Stability | Highly Reactive (Oxidation) | Stable (Protected by Liposomes) |
Market Positioning | Economy / Mass Market | Premium / Clinical Grade |
Why Brand Decision-Makers are Choosing LiposoMore

For brand owners and manufacturers, the switch to LiposoMore’s Liposomal Iron isn't just a scientific upgrade—it’s a strategic business move.
Market Differentiation: Offering a "Stomach-Friendly" or "High-Absorption" claim allows for premium pricing and higher margins.
Formulation Versatility: Our liposomal technology ensures stability in various delivery formats, including liquid sachets, capsules, and functional gummies, without the risk of oxidation or off-notes.
Proven Compliance: When consumers feel the results without the side effects, brand loyalty increases, leading to higher re-order rates.
Specification of LiposoMore Liposomal Iron (Ferric Pyrophosphate)
Items | Specification | Test Method |
Appearance | White to light yellow powder, free of peculiar smell, odor, corruption and mildew, and visible foreign impurities | USP <631> |
Odor | Odorless | USP <631> |
Solubility | Dispensible in water | USP <631> |
Iron Purity | 7%-9% | USP <730> |
Nanoparticle size | 100-300nm | DLS |
Encapsulation efficiency | ≥80% | In-house |
Loss on Drying | ≤10.0% | USP <731> |
Total Heavy Metal | ≤10ppm | USP <231> / ICP-MS |
Lead (Pb) | ≤3ppm | USP <232> ICP-MS |
Mercury (Hg) | ≤0.1ppm | USP <232> ICP-MS |
Cadmium (Cd) | ≤1.0ppm | USP <232> ICP-MS |
Arsenic (As) | ≤1.0ppm | USP <232> ICP-MS |
Total Plate Count | ≤1000cfu/g | USP <61> |
Molds & Yeasts | ≤100cfu/g | USP <61> |
E.Coli | Negative/gram | USP <62> |
Salmonella | Negative in 25 gram | USP <62> |
Staphylococcus Aureus | Negative in 25 gram | USP <62> |
Solving the "Metallic Challenge" in Pediatric Liquid Drops

To understand the transformative power of LiposoMore, let’s look at one of the most difficult formulations in the nutraceutical industry: Pediatric Iron Drops.
The Challenge
A leading European supplement brand approached us with a common R&D crisis. Their existing Ferrous Sulfate liquid drops were failing in the market. Despite a high-potency formula, the product had three fatal flaws:
1. Palatability: The intense metallic aftertaste led to poor compliance in infants (spit-up).
2. Stability: Within three months of shelf life, the liquid would oxidize, turning from a clear solution to a murky, brown-tinted suspension with a rancid smell.
3. Teeth Staining: Parents complained that the direct contact of iron salts was causing dark stains on their children's developing teeth.
The LiposoMore Solution
The brand reformulated using LiposoMore’s Liposomal Ferric Pyrophosphate specifically designed for aqueous dispersion.
Because our ferric pyrophosphate is fully "shrouded" within a lipid bilayer, the iron molecules never make physical contact with the taste buds or the tooth enamel.
The Result
Zero Metallic Taste: The R&D team achieved a "Clean Label" profile, using only natural fruit extracts for flavoring without needing heavy sweeteners to mask the iron.
Superior Stability: Our liposomes remained intact in the liquid base, preventing oxidation. The product maintained its color, odor, and potency throughout its 24-month accelerated stability testing.
Market Success: Upon relaunch, the brand reported a decrease in consumer complaints related to taste and a  increase in repeat purchases from parents.
increase in repeat purchases from parents.
"By switching to LiposoMore, we transformed a 'functional but unpleasant' product into a market leader. It allowed us to claim 'Stomach-Friendly' and 'No Teeth Staining' on the front of the box—two massive wins for our marketing team." — Head of R&D.
Conclusion: The Future of Iron Supplementation
The "cheap but ineffective" era of iron supplementation is ending. For R&D teams looking to innovate and procurement managers aiming to future-proof their product lines, Liposomal Iron represents the most viable path forward.
LiposoMore provides the technical expertise and high-purity ingredients needed to turn these scientific advantages into market-leading products.
Ready to Upgrade Your Product Line?
Contact the LiposoMore Technical Team today to request a sample or download our full Clinical Comparison Whitepaper. Let’s build the next generation of iron supplements together.
FAQs about Liposomal Iron Supplementation
1. What are the main advantages of liposomal iron compared to ferrous sulfate?
The main liposomal iron benefits include significantly higher bioavailability, reduced gastrointestinal irritation, and improved stability in formulations. Unlike ferrous sulfate, liposomal iron is encapsulated in a phospholipid layer, allowing it to bypass traditional absorption barriers and minimize side effects such as nausea and constipation.
2. Is liposomal iron suitable for sensitive groups such as children or pregnant women?
Yes, liposomal iron supplement formulations are widely considered suitable for sensitive populations due to their gentle, non-irritating properties. The liposomal structure helps prevent direct contact with the stomach lining, reducing common discomfort and making it a preferred choice in pediatric and prenatal applications.
3. How does liposomal iron improve absorption in the body?
Unlike traditional iron salts, liposomal iron is absorbed through alternative pathways such as endocytosis and the lymphatic system. This mechanism avoids common absorption inhibitors in the digestive tract, which is one of the key liposomal iron benefits contributing to higher iron uptake and improved efficacy.
4. Why are more brands switching to liposomal iron supplements?
Brands are increasingly adopting liposomal iron supplement solutions because they offer a strong competitive advantage. With claims such as “high absorption” and “stomach-friendly,” companies can differentiate their products, improve customer satisfaction, and achieve higher repeat purchase rates.
[Contact Us] | [Request a Technical Datasheet]
Natalie Wu
Hey Guys, I am Natalie Wu, an R&D researcher at LiposoMore. I graduate from College with a Master’s Degree in Bioengineering, I would like to share news and the science behind liposomal ingredients. At LiposoMore, we drive ingredient development with scientific precision and a passion for advanced nutritional technologies.

 EN
EN
 jp
jp  ko
ko  fr
fr  de
de  es
es  ru
ru  pt
pt  ar
ar  pl
pl  vi
vi 



































